Protein Helps Cells Handle Stress
Key Takeaways:
- Protein Dynamics: Under stress, the protein Dsk2 changes its structure to connect with nearby molecules, forming droplet-like clusters that help manage damaged proteins.
- Collaborative Studies: Studies from Syracuse and the University of Pittsburgh, published in The EMBO Journal, demonstrate that this mechanism works similarly in both yeast and humans.
- Link to Disease: Mutations associated with ALS interfere with the formation of these clusters, suggesting a potential failure in the cell’s protein recycling system.
Inside every cell, a cleanup operation runs around the clock. Proteins are constantly damaged by wear and tear. Some can be repaired, while others must be dismantled and recycled. When this system fails, damaged proteins accumulate in clumps associated with diseases such as Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Dementia.
A new study published in The EMBO Journal by Nirbhik Acharya (postdoc) and Carlos Castañeda, associate professor of biology and chemistry at Syracuse University’s College of Arts & Sciences (A&S), reveals a key part of how that cleanup system works and what goes wrong in disease.
How the sorting hubs form
Castañeda's team worked with Dsk2, a yeast protein that functions similarly to human ubiquilin-2, which shuttles damaged proteins to the cell’s recycling machinery. When that shuttling breaks down, damaged proteins can build up, a hallmark of ALS.
“Dsk2 offers a window into the basic rules that guide how this system operates across species,” says Castañeda.
To track those rules in action, the team used nuclear magnetic resonance spectroscopy — essentially an MRI for individual molecules — which lets scientists observe subtle structural changes on an atomic level as a protein moves and interacts with other molecules.
What they found was striking. Under stress, Dsk2 reshapes itself and links with neighboring molecules to form biomolecular condensates. These are temporary, droplet-like clusters where damaged proteins are gathered and potentially processed. Because these clusters assemble and dissolve through many small, reversible interactions, the cell can build or break them apart on demand.
At the heart of this mechanism is a folded region called the STI1 domain, shaped like a clamp with a groove. Short spiral segments elsewhere in the protein slip in and out of this groove, binding briefly, then releasing. This process allows different parts of the same molecule to interact and enables multiple Dsk2 molecules to link together into clusters. The importance of this architecture became clear when the researchers removed Dsk2, or just its spiral segments. Cells struggled to form clusters potentially leading to dysfunctional protein quality control.
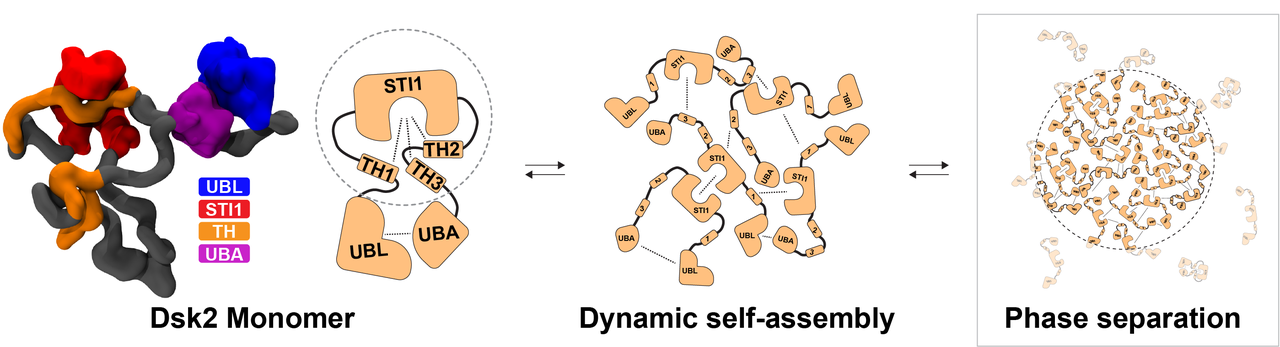
The study reveals, for the first time, how specific structural features within Dsk2 drive the formation of these condensates in living cells.
Together, four teams connected molecular structure, simulated behavior, and real-world cell responses. Castañeda’s data were complemented by computer simulations from Shahar Sukenik’s lab in Syracuse’s Department of Chemistry, where postdoctoral researcher Jessica Niblo helped model how Dsk2 behaves, and by cell-based experiments from Jeroen Roelofs and his PhD student Emily Daniel at the University of Kansas Medical Center, who tested how changes in Dsk2 affect real yeast cells under stress. Work with Daniel Kraut’s lab at Villanova University reconstituted Dsk2 clusters with protein recycling machinery.
A coordinated finding
Those yeast-cell results gain further weight from a parallel set of experiments. While Castañeda's team studied the mechanism in living yeast cells, collaborators led by Matthew Wohlever at the University of Pittsburgh used X-ray crystallography to capture the first detailed structural snapshot of a ubiquilin STI1 domain. The researchers found that mutations associated with ALS interfere with how the STI1 clamp works, suggesting a potential failure in the cell’s protein recycling system.
The two studies, published back-to-back in The EMBO Journal, offer complementary views of the same mechanism. The yeast work shows how it unfolds inside living cells; the structural work reveals the molecular contacts that make it possible. Taken together, they suggest this system is a fundamental strategy that cells rely on.
Castañeda and Wohlever had discussed preliminary data at an American Society for Biochemistry and Molecular Biology annual meeting. “We realized that we saw a similar molecular mechanism working in both yeast and human versions of ubiquilin,” says Castañeda. “We had an ‘a-ha’ moment that pushed both projects forward.”
What comes next
These findings sharpen the picture of how a cell’s protein-recycling system is organized, and how subtle disruptions to that organization may contribute to disease. For scientists studying neurodegeneration, the work offers an important step toward understanding what happens when the cell’s cleanup crew stops functioning as it should.
“We’re starting to understand the rules,” says Castañeda.
A future challenge, he suggests, is learning how to intervene when those rules break down. Because similar protein-clumping problems occur across many neurodegenerative diseases, this mechanism may represent a common target for future therapies.
Published: April 2, 2026
Media Contact: asnews@syr.edu
